 Heavy mesons, such as pions and pi-mesons form the short-range version of the strong nuclear force. The interaction between these particles is powerful and has an exquisitely fine-tuned nature-neutrons, the lightest strongly interacting particles, exchange mesons with protons. Neutrons and protons exchange mesons to form a strong nuclear force. Because protons must be close to each other, a strong nuclear force is necessary to hold two nucleons together. These exchanges of mesons are like the constant hitting of tennis or ping pong balls. Because the protons must be close to each other to interact, they must exchange mesons. Neutrons and protons can be found in all atoms, and each proton has a positive charge. The strong nuclear force is a powerful attraction between two charged particles. They exchange mesons with protons to form a strong nuclear force. While they are roughly the same as protons in an atom, their mass is nearly half that of a proton. In simpler versions, electrons are seen as particles orbiting the nucleus, but in reality, they are much more complex. Because the positive and negative charges on each particle attract each other, electrons move through space. In atoms, neutrons and protons are found inside the nucleus, which is the center of an atom. Therefore, scientists are constantly working on finding ways to make this difference clearer. However, it may not be evident to an outside observer. This difference in composition is fundamental to atomic physics. The interior of a neutron is similar to that of a proton, although neutrons contain different types of quarks. They would be much heavier than one another if they had the same mass.Ī neutron’s and proton’s mass is the same, but their properties differ. 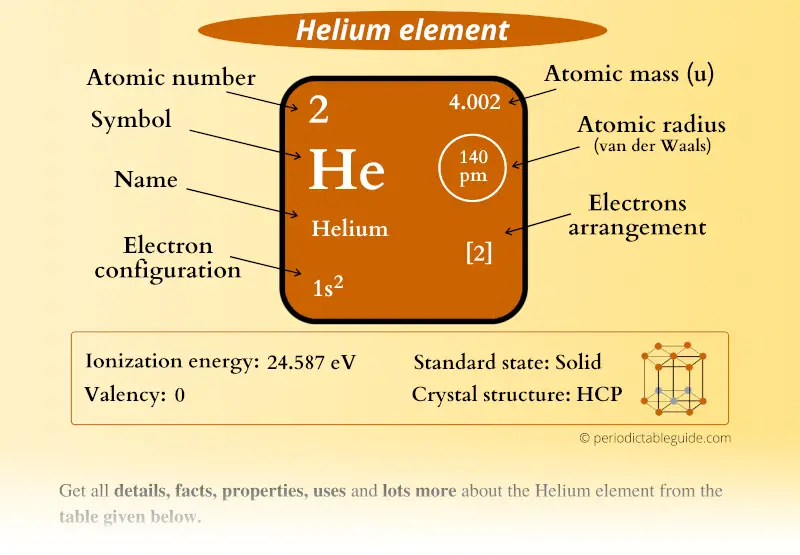
While protons and electrons are similar in appearance, they are significantly different in mass and charge. This difference between protons and electrons makes it easy to see how they differ. The mass of a neutron is only 0.054% of the mass of a proton. Protons and neutrons have equal masses, but electrons are lighter. In this way, the net charge is positive, which means that there is one more electron than a molecule’s mass. In this case, the amount of electrons is two times greater than the number of protons. An atom can also be characterized by its number of electrons. However, they are bonded with the protons with strong residual force. Unlike protons, neutrons have no net electrical charge. Because the electron and proton are the same sizes, their charge cancels each other. In an atom with only neutrons, the charge on a neutron is negative, so it is neutral. 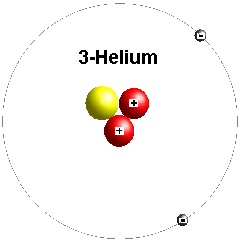
However, the middle area had a higher charge than the interior and exterior. This made the proton positively charged throughout. The proton has two electrons and one down quark. Neutral atoms have the same number of protons and electrons, and the overall charge is neutral. The negative charge of electrons contributes to the charge of an atom. Neutrons have a net negative charge, while protons are positively charged. These atoms are in equilibrium when they have an equal number of protons and electrons. 
What is an atom? Atoms are small units of matter that contain protons and electrons. Protons comprise almost all of the mass of an atom. Their mass is one atomic mass unit (amu), or 1.67 kilograms. Protons are charged positively with a positive electric charge of one (+1). Protons are located in the nucleus, the tiny dense region at the center of the atom. In an atom, there are three particles: Protons, Neutrons, and Electrons. Where Are Neutrons and Protons Located in an Atom? 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
